Pertussis - HAN
Jump To:
Overview
The bacteria are shed in nasopharyngeal secretions and can be spread through coughing, sneezing, or direct contact with fluids from the nose, mouth, or throat of an infected person. If untreated, infected individuals can spread the pertussis bacteria to others from 1 week before cough onset to 21 days after cough onset. Untreated infants (<1 year) remain infectious for up to 42 days from cough onset. When treated with antibiotics, the infectious period is reduced to 5 days after the initiation of treatment. Severe infections can result in complications such as pneumonia among all age groups. Seizures and encephalopathy generally occur only among infants. The average incubation period for pertussis is 7-10 days with a range of 4-21 days.
Case Definition
A cough illness lasting ≥ 2 weeks with at least 1 of the following signs or symptoms:
Paroxysms of coughing, OR
Inspiratory “whoop,” OR
Post-tussive vomiting, OR
Apnea (with or without cyanosis) (for infants <1 year of age only)
Diagnostics Testing
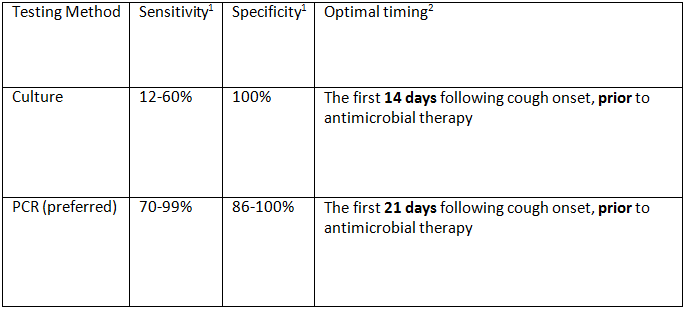
- Locally available FDA approved PCR tests for B. pertussis
- Multiplex assay: Biofire FilmArray Respiratory Panel, bioMérieux
- Targets the pertussis toxin promoter (ptxP): highly specific
- Multiplex assay: Biofire FilmArray Respiratory Panel, bioMérieux
- Other locally available PCR tests for B. Pertussis
- Singleplex assay: Typically, an in-house laboratory developed PCR method
- Targets the insertion sequence IS481: highly sensitive
- It is important to note research has found that PCR can detect pertussis in infants less than 6 months of age up to 3 weeks following pertussis treatment initiation. Pertussis positive PCR results may correspond to the presence of antibiotic-damaged bacteria. As a result, the clinical significance of pertussis detected by PCR after initiation of antibiotic treatment is unclear. For more information, see Real-Time PCR Measurement of Persistence of Bordetella pertussis DNA.
Bordetella pertussis Commercial Lab Reference Table
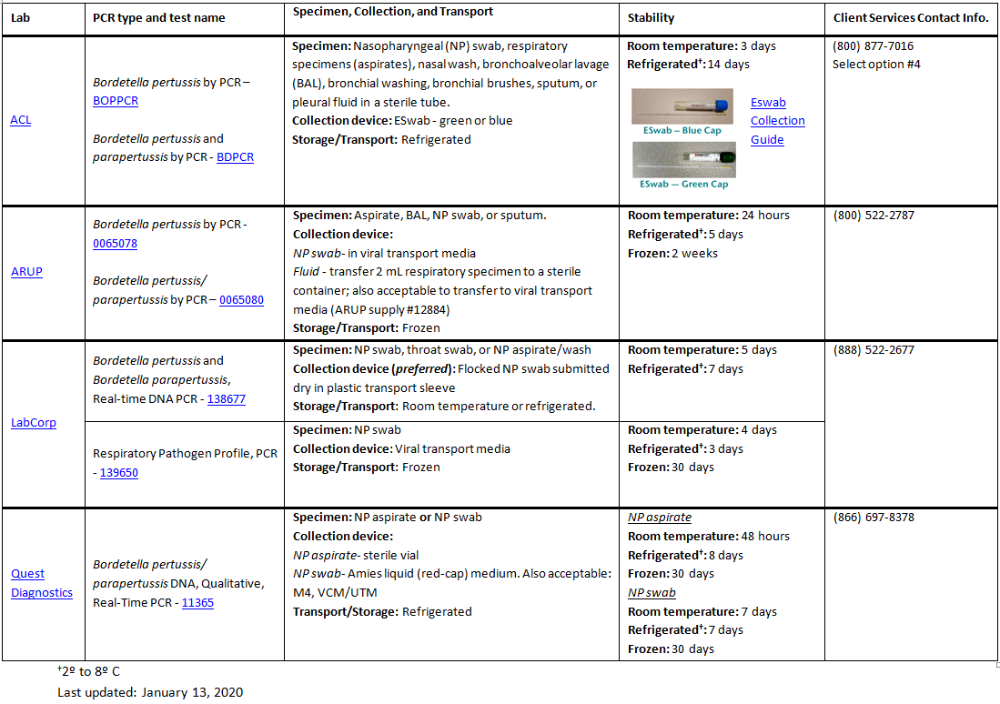
Specimen Collection
Collect nasopharyngeal (NP) swabs as soon as pertussis is suspected, within 21 days of cough onset.
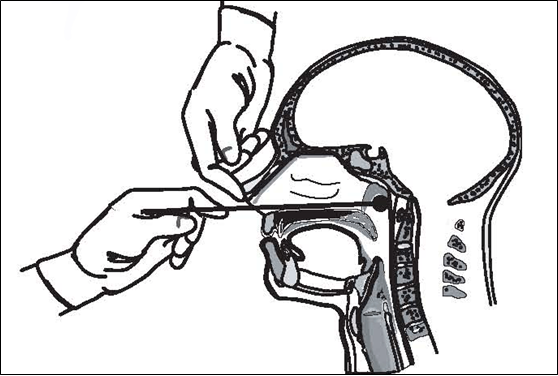
- Carefully insert swab through the nares straight back (not upwards) until reaching the posterior wall of the nasopharynx.
- Gently roll and rub the swab.
- Leave the swab in place for several seconds before slowly removing the swab.
- For Specimen Handling, see CDC Specimen Collection Guidelines.
Prenatal Care To Prevent Pertussis
Information for Vaccinators
Additional Resources
CDC: Manual for the Surveillance of Vaccine-Preventable Diseases; Chapter 10: Pertussis
CDC’s Pertussis Vaccination Website
CDPH HAN Alert Pertussis Clinical Guidelines for Testing and Treatment of Persons with Pertussis
CDC: Pertussis Specimen Collection
IDPH: Pertussis
CDPH: Pertussis Reporting for Schools
Clinical Guidelines for Mgmt of HCP Exposed to Pertussis
Update: Increased Pertussis Activity November 20, 2019
To report suspect or confirmed cases:
Do not wait for laboratory confirmation.
- If you have access to I-NEDSS (Illinois’ National Electronic Disease Surveillance System), please report via that electronic system.
- Otherwise, use the Online Case Reporting Form (preferred) OR contact the CDPH Disease Reporting Hotline at 312-743-9000, option 2.*
Questions:
For Questions: Contact the CDPH Disease Reporting Hotline at 312-743-9000, option 2.Schools: Please click here for reporting and management information.
Pertussis Contact
Complete the Online Case Report Form (*preferred) or
Fax paper Case Report Form to:
(312) 746-6388 or
Call Provider Reporting Hotline:
(312) 743-9000
Sign up for electronic reporting through the Illinois National Electronic Disease & Surveillance System (I-NEDSS)
For questions during non-business hours call 311.
(312-744-5000 if outside the City of Chicago.)
